Teva Pharmaceuticals to pay New York $523 million to settle claims from its role in opioids crisis - ABC News

Teva Pharmaceuticals USA Issues Voluntary Nationwide Recall of all Amlodipine/Valsartan Combination Tablets and Amlodipine/Valsartan/Hydrochlorothiazide Combination Tablets that are Within Expiry | FDA
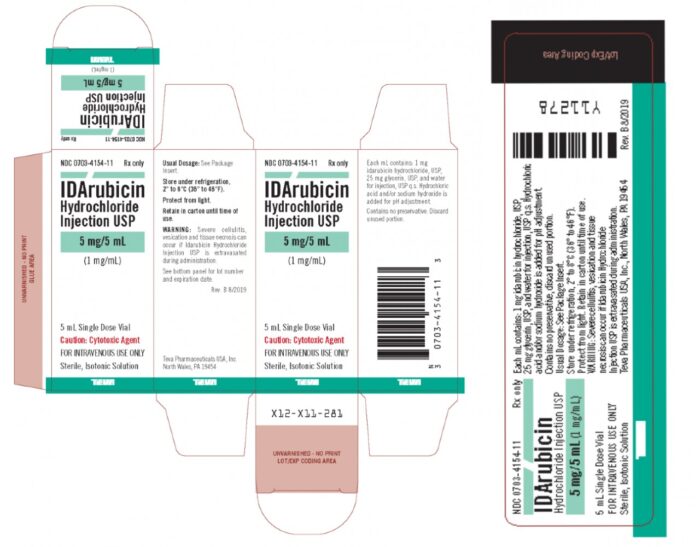
Teva Pharmaceuticals Recalls Acute Myeloid Leukemia Drug Over Particulate Matter Contamination - Top Class Actions
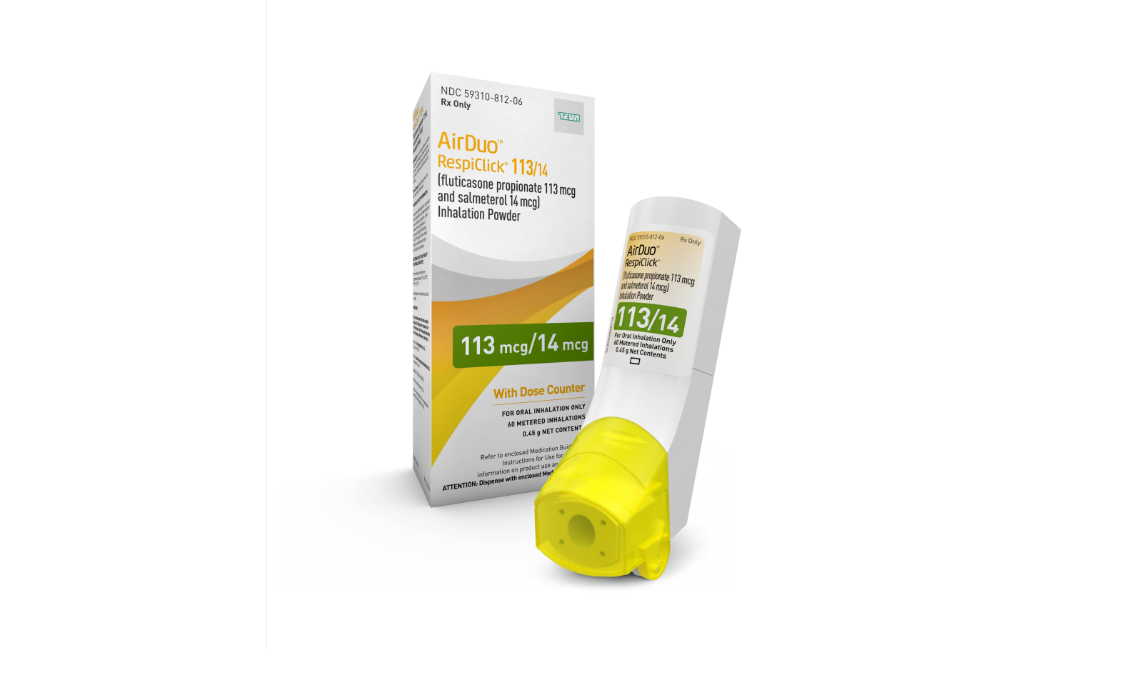
Teva Introduces Inhalation Powder and Generic Version in U.S. for Asthma Treatment - Drug Discovery and Development

Teva Pharmaceuticals found to be responsible for its role in the opioid epidemic in New York State | CNN

Teva Pharmaceuticals and IBM Expand Global Partnership to Enable Drug Development and Chronic Disease Management with Watson